Life
Sciences
We provide timely and accurate CDMO/CRO services to our customers throughout the drug development process.
improving our health
We believe the therapies our customers develop are critical to improving lives and we are proud to be a part.
Pace® Life Sciences Announces Successful Outcomes Following US FDA Inspection of Operations in San German, Puerto Rico
Pace® Life Sciences announced the receipt of an overall positive review from The Food & Drug Administration (FDA)
Pace® Life Sciences Appoints Dawn Von Rohr as Division President
Von Rohr is an experienced leader with over 25 years of expertise in global CDMO strategic growth initiatives and operational improvements, specifically within the pharmaceutical industry.
Explore our range of services
Wherever you are in your drug development journey, we can help.
- Characterization of Novel Molecules & Biologics
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Characterization of Novel Molecules & Biologics
- Test Article Preparation
- Formulation Development
- Lyophilization/Spray Drying/Hot Melt Extrusion/Milling
- Analytical Development
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Regulatory Consulting
- Test Article Preparation
- Formulation Development
- Lyophilization/Spray Drying/Hot Melt Extrusion/Milling
- Analytical Development
- GMP Clinical Trial Manufacturing
- Reference Standard Programs
- Microbiology
- Analytical Instrumentation Services
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Test Article Preparation
- Formulation Development
- Lyophilization/Spray Drying/Hot Melt Extrusion/Milling
- Analytical Development
- Regulatory Consulting
- GMP Clinical Trial Manufacturing
- Reference Standard Programs
- Microbiology
- Cleanroom Certification
- Extractable/Leachable Studies
- ICH Stability Programs
- Formulation Development
- Lyophilization/Spray Drying/Hot Melt Extrusion/Milling
- Analytical Development
- GMP Clinical Trial Manufacturing
- Reference Standard Programs
- Microbiology
- Extractable/Leachable Studies
- ICH Stability Programs
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Regulatory Consulting
- Cleanroom Certification
- Mapping Services
- Facility & Equipment Validation
- Formulation Development
- Lyophilization/Spray Drying/Hot Melt Extrusion/Milling
- Analytical Development
- Reference Standard Programs
- Microbiology
- Extractable/Leachable Studies
- ICH Stability Programs
- Raw Materials Clearance
- Finished Product
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Regulatory Consulting
- Cleanroom Certification
- Mapping Services
- Facility & Equipment Validation
- Environmental Monitoring
- Reference Standard Programs
- Microbiology
- Extractable/Leachable Studies
- ICH Stability Programs
- Raw Materials Clearance
- Finished Product
- Analytical Instrumentation Services
- Laboratory Relocation Services
- Scientific Staffing
- Cleanroom Certification
- Mapping Services
- Facility & Equipment Validation
- Environmental Monitoring
THE RIGHT PARTNER FOR YOUR PROJECT
We have a nationwide network of state-of-the-art facilities, each with long-established histories of successful product development and commercialization and excellent audit outcomes from regulatory agency and client reviews.
FDA
REGISTERED
DEA
REGISTERED
CLASS (I-IV)
GMP
COMPLIANT
ISO
17025
ACCREDITED
OUR PROMISE TO YOU
Our investment in state-of-the-art facilities and highly trained experts emphasizes our commitment to delivering positive customer experiences across all phases of pharmaceutical and biopharmaceutical development.
- RELIABLE DELIVERY
- COLLABORATIVE RELATIONSHIPS
- EXCEPTIONAL SERVICE
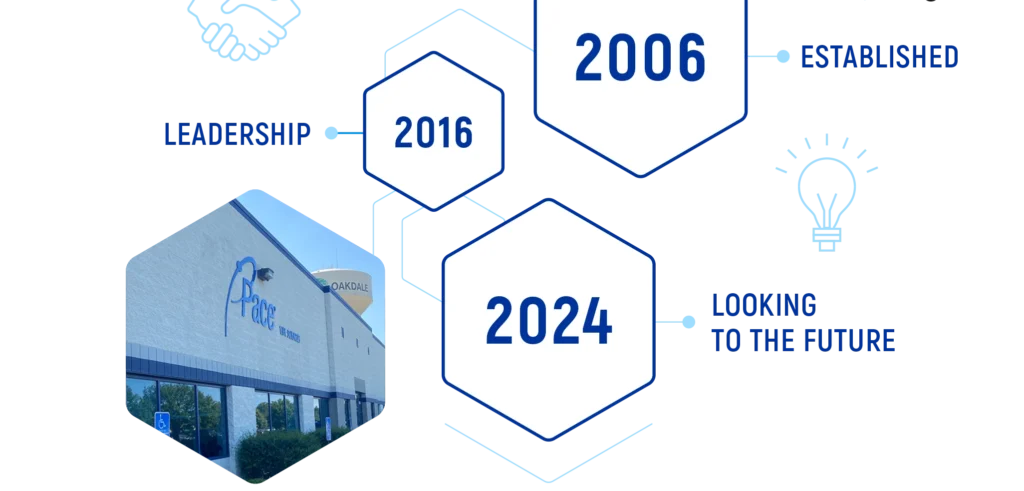
IT ALL STARTED IN 2006
Since 2006, Pace® Life Sciences has continued to prioritize strategic investments and acquisitions to meet the changing needs of our customers. As the market changes, we are committed to making sure we are positioned as the best end-to-end solution for your program.
Conferences

LNP Formulation & Process Development Summit
Fast Track Your End-to-End LNP Development, Entering into Gene Therapy to Vaccination
& Beyond with Safety & IP Confidence for Accelerated Approval
April 29 – May 2, 2024 | Boston, MA
feature webinar

Navigating the Investigational New Drug (IND) Application Process
Life Sciences, Clinical Trials, Pharmaceutical Regulation, Pharmaceutical, Drug Discovery & Development, Drug Safety.
Wednesday, March 06, 2024 | 1pm EST (12pm CST / 10am PST)
60 min




